Items list
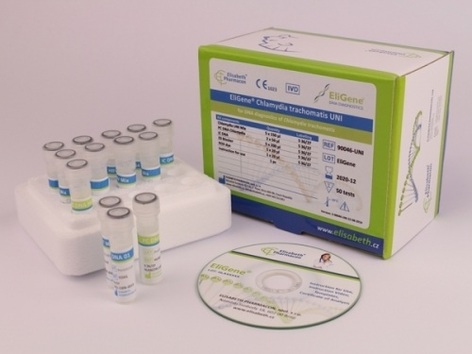
EliGene® Chlamydia Trachomatis
EliGene® Neisseria
EliGene® Ureaplasma
EliGene® Mycoplasma hom/gen
EliGene® Ureaplasma/Mycoplasma
EliGene® detection kits- The usage of the Internal Control allows to monitor the extraction procedure and to check possible PCR inhibition
- Kits are supplied with positive control allowing quality control of the analysis
- Simultaneous amplification (multiplex) of DNA/RNA from pathogen and internal control in one PCR tube
- The usage of Hot-Start technology minimize the risk of non-specific reaction and provide ultimate sensitivity 1-10 copies of nucleic acid/reaction
- The usage of Ready to use Mastermixes containing all reaction components for easier reaction setup and handling
- The usage of uracil in the Mastermixes of UNI kits enables UNG treatment to prevent the re-amplification of carryover-PCR products by removing any uracil incorporated into single- or double-stranded DNA
- vTo ensure the highest quality of our kits, the row material from the leading providers is used
- Quantitative Standards in the most of the kits enables to calculate precisely the NA load from 102 to 106 copies/reaction
- High stability of used reagents
- The usage of EliGene® UNI kits enables complete detection in 50 minutes
- Kits are CE certified IVD
- Kits are validated on LightCycler® 1.2, 2.0, Nano and 480, ABI® 7300, 7500 and 7500FAST, RotorGene-Q (RotorGene 6000)
- Compatibility of the kits with broad range of instruments - RotorGene®, SmartCycler®, Mx3000P® and 3005P® QPCR systems or iQ5®, CFX96® and other cyclers